
It is a depiction of the periodic law, which says that when the elements are arranged in order of their atomic numbers an. 
An elements placement on the periodic table indicates whether its chemical charge is negative or. It is an icon of chemistry and is widely used in physics and other sciences. A chemical charge can be found by using the periodic table. For example, Both O 2-, Mg 2+ have 10 electrons but they don’t have the same ionic radius as the effective nuclear charge in both of them is. 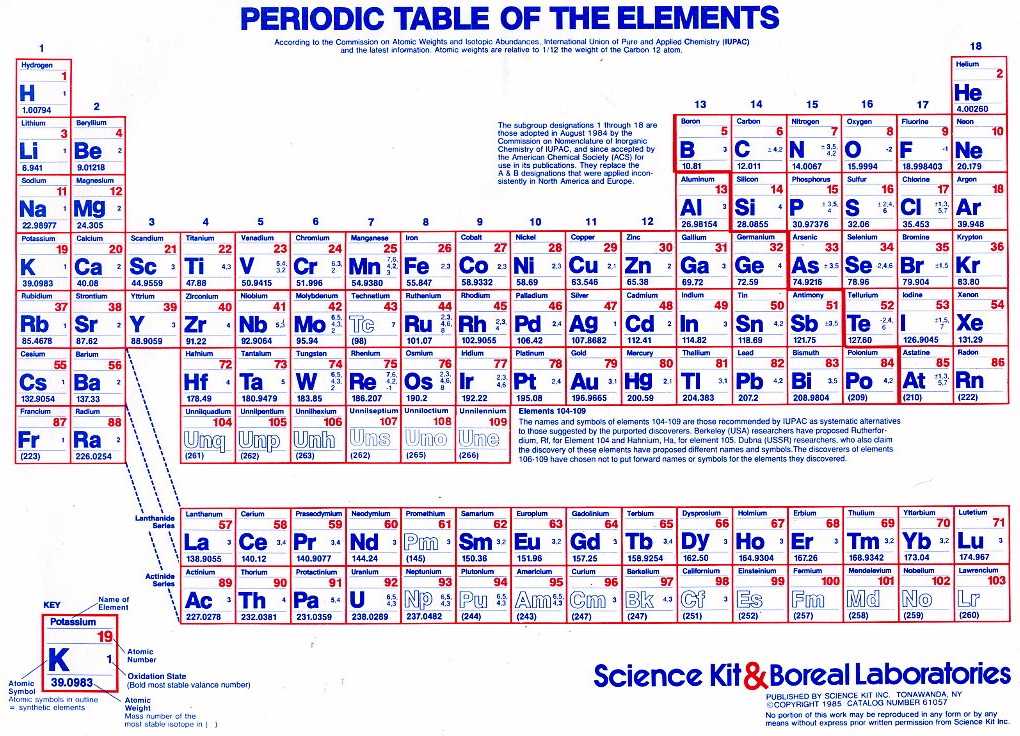
An iron ion with a charge of +8 is not very likely, therefore, the octet rule is not applicable to transition elements. The periodic table, also known as the periodic table of the elements, arranges the chemical elements into rows ('periods') and columns ('groups'). Atoms and ions which consist of an equal number of electrons are considered isoelectronic species. All the metallic elements located on the left part of. In order for an element such as iron (Fe) to achieve the same noble gas configuration of argon (Ar), it would need to lose 6 electrons in the 3 d subshell and 2 electrons in the 4s subshell. acetate PERIODIC TABLE OF IONS arsenate arsenite benzoate borate bromate carbonate chlorate chloride chlorite chromate cyanate cyanide dichromate CH3COO AsO4 3 AsO3 3 C6H5COO BO3 3 BrO3 CO 3 2 ClO3 Cl ClO2 CrO4 2 CNO CN Cr2O7 2 oxalate perchlorate periodate permanganate peroxide phosphate. There is also a very clear way of knowing whether an element has a positive or a negative ionic charge. acetate PERIODIC TABLE OF IONS arsenate arsenite benzoate borate bromate carbonate chlorate chlorite chromate cyanate cyanide dichromate CH3COO AsO4 3 AsO3 3 C6H5COO BO3 3 BrO3 CO3 2 ClO3 ClO2 CrO4 2 CNO CN Cr2O7 2 oxalate perchlorate periodate permanganate peroxide phosphate pyrophosphate sulfate. This is because the transition metals have electrons in d subshell and do not follow the octet rule. The image below shows the general trend for ionic charge. Elements on the left side of the periodic table, metals, lose the electrons necessary to reach the electron configuration of the nearest noble gas. Thus, the periodic table becomes a tool for remembering the charges on many ions. But you need to know the specific ionic charge elments. Non-metals (found on the right) will be negative. Note that CuO has only one copper ion, while Cu 2 O has two copper ions. Based on position on the periodic table, the oxide ion has a charge of 2. On the Periodic Table metals (found on the left of the table) will be positive. Write the name leaving room for a Roman numeral, since copper forms ions with variable charges. For example, iron(II) has a 2+ charge iron(III) a 3+ charge. Notice that there is no simple pattern for transition metal ions (or for the larger main group elements) as there is with the main group ions. To find the ionic charge of an element you'll need to consult your Periodic Table. Roman numeral notation indicates charge of ion when element commonly forms more than one ion. The covalent radius of Cl 2 is half the distance between the two chlorine atoms in a single molecule of Cl 2.\) shows the characteristic charges for some of these ions. ion charge stock name (IUPAC) 58 Ce3+ cerium 59 Pr3+ praseodymium 60 Nd3+ neodymium 61 Pm3+ promethium Sm 2+ samium(II) Eu2+ europium(II) 64 Gd3+ gadolinium 65 Tb3+ terbium 66 D圓+ dysprosium 67 Ho3+ holmium 68 Er3+ erbium 69 Tm3+ thulium Yb 2+ ytterbium (II) 71 Lu lutetium 91 Pa5+ protactinium (V) 92 U6+ uranium (VI) 94 Pu4+ plutonium (IV) 95. predict which element differs the most from. predict which element forms the most stable compounds in the +2 oxidation state. (d) This is a depiction of covalent versus van der Waals radii of chlorine. Based on the positions of the group 14 elements C, Si, Ge, Sn, and Pb in the periodic table and the general trends outlined in this section, classify these elements as metals, semimetals, or nonmetals. (c) The van der Waals atomic radius, r vdW, is half the distance between the nuclei of two like atoms, such as argon, that are closely packed but not bonded. (b) The metallic atomic radius, r met, is half the distance between the nuclei of two adjacent atoms in a pure solid metal, such as aluminum. 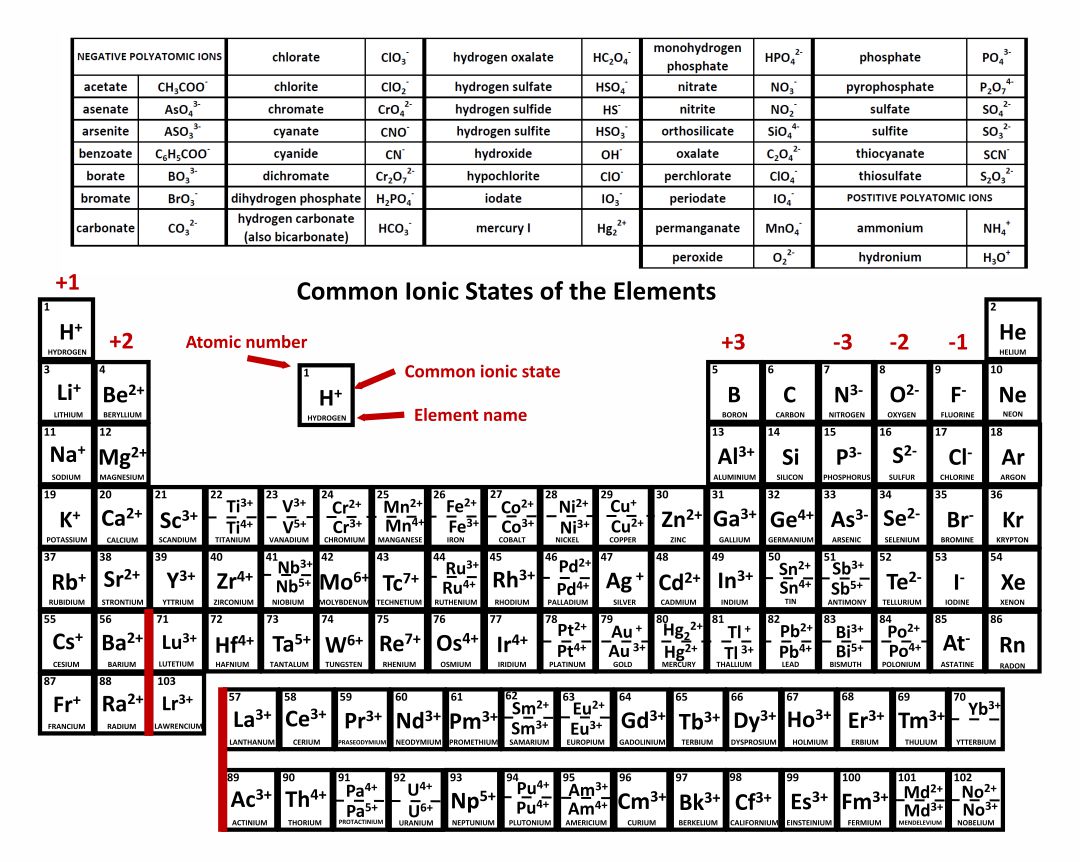
(a) The covalent atomic radius, r cov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as Cl 2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
